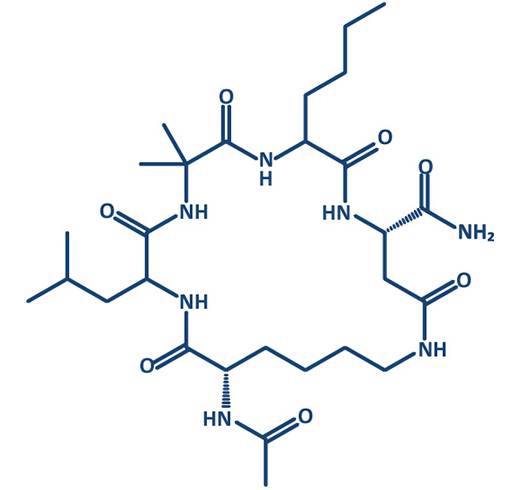
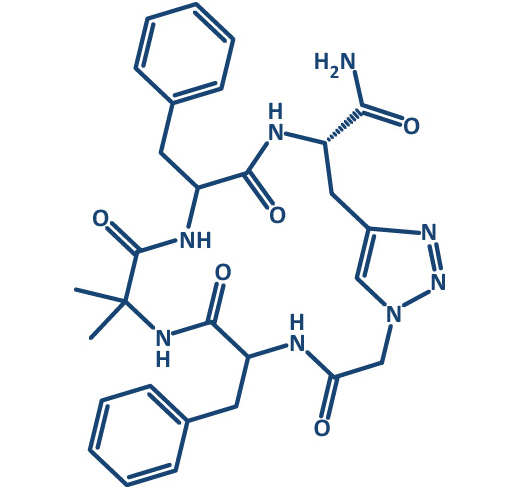
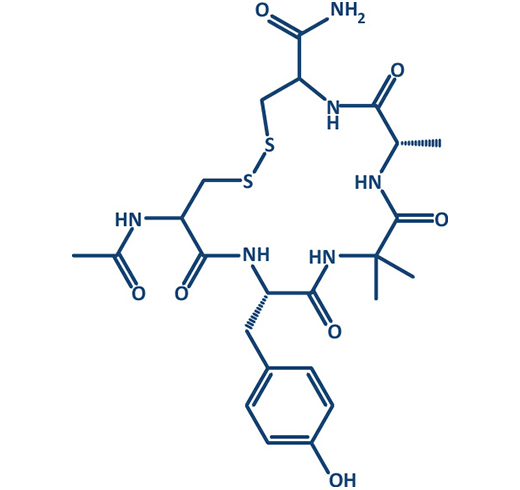
In the past decade, interest into macrocycles strongly increased in medicinal chemistry. More than 100 macrocyclic drugs and clinical candidates are currently marketed or in drug discovery programs.
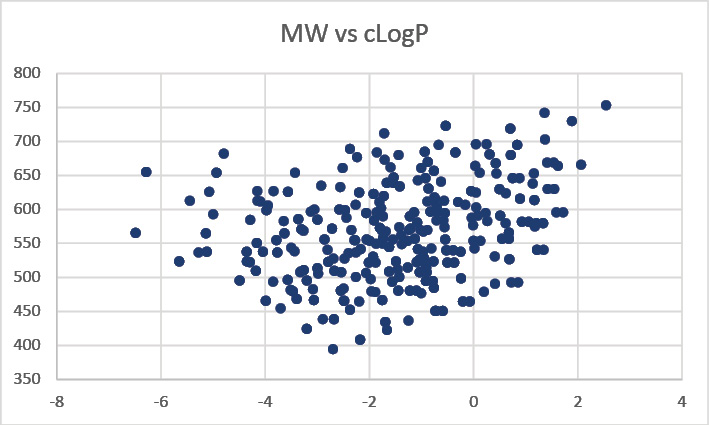
Prestwick Chemical Libraries in collaboration with GENEPEP include a screening library of 400 high purity short macrocyclic peptides designed with a mix of natural and non-natural amino acids and different types of cyclization (disulfide, lactam, triazole). Macrocyclization can provide a method of improving target potency and modulating selectivity. Macrocycles can become more drug-like than expected based on their physicochemical and pharmacokinetic properties.
Moreover, cyclic peptides in comparison to their linear counterparts combine multiple interesting properties like low toxicity and improved enzymatic stability; they are metabolically more stable, and should have a lower degree of flexibility, with more defined secondary structure conformations that can be adapted to imitate protein interface.